Design in healthcare, aviation or finance projects can seem restrictive due to the regulation employed in these fields. At Solita, we carry out a lot of meaningful projects for demanding and regulated environments. Read how regulation can make the design of a medical device, for example, particularly interesting.
Building and improving digital services related to health and well-being seems to be a meaningful design challenge for many, with a good purpose . At least for me, the work has felt meaningful when I have been able to participate in planning healthcare services for both the public and private sector at Solita. At the same time, solving healthcare design problems is particularly challenging. This is indicated by the challenges of launching the social and healthcare reform and well-being services counties in Finland, the strain and mental health crisis that followed the COVID-19 pandemic, the complexity of the sector’s fragmented information systems, as well as the regulation related to it. In this blog article, I want to talk specifically about the regulation of medical devices and the opportunities it offers to designers.
The rewarding nature of designing medical devices comes from the following themes, which are widely appreciated in Solita’s design community:
- A human-centred approach
- Continuous development and improvement
- Sustainable design and effectiveness
- Holisticity and working in a multi-skilled team
My point of view and my own work experience with medical devices is strongly focused on online services and software instead of concrete devices. That is why I often refer to them with the catchy word “service” in this article. More specifically, I am talking about Class II a devices according to the MDR, which are less risky in terms of patient safety than, for example, higher Class II b or III devices installed in the body. (You can read more about the meaning of the various classes e.g. on Fimea’s website.)
A people-centred approach written into the design
For a designer interested in combining human understanding and technology, the usability standard and its technical report serve as a guide to manufacturing medical devices. So in the language of standards: IEC-62366-1:2015 and IEC-62366-2:2016. In addition, the design of medical devices is essentially regulated by the Medical Device Regulation (MDR), as well as the related quality, risk management and software development standards. At first glance, design work tied to regulation may seem difficult, but I personally see the usability standard as an advocate of human-centred design that sells and justifies the principles of user-centred design for software development “on behalf of the designer”. The medical device must meet the needs of the users, be patient-safe for its intended use and be easy to use so that serious hazarduous situations cannot arise from its use. Could you ask for a better “defence speech”?
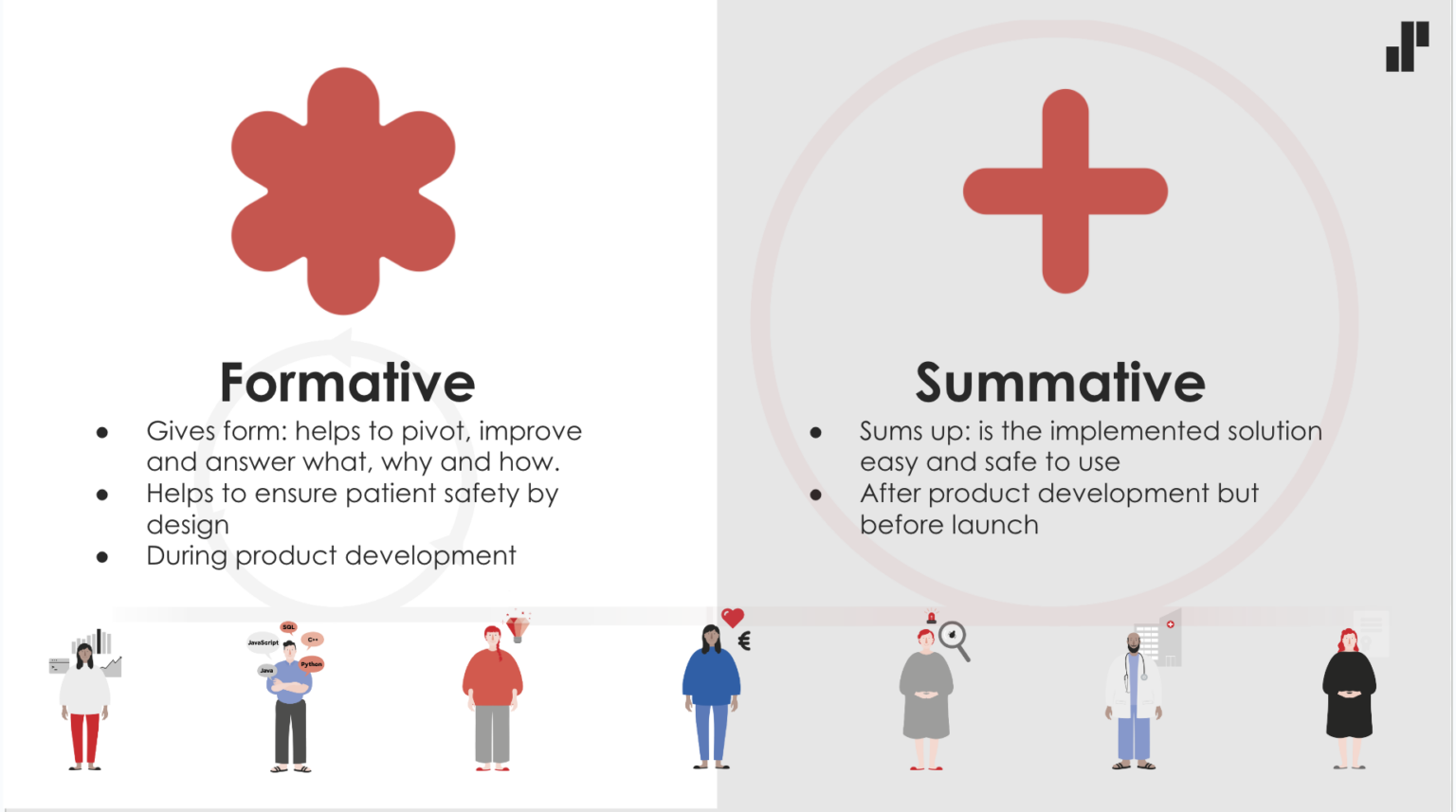
The usability standard both requires and gives the freedom to shape a service utilising different kinds of participatory methods throughout the entire product development process. In the language of the standard, this human-centred design with end users and stakeholders is called formative evaluation. In addition, the standard requires that, before release, the ease of use of the service and patient safety are ensured by an impartial summative evaluation. Summative evaluation usually means usability testing with end users in as authentic an environment as possible, involving 10-15 users per target group. The usability standard for medical devices puts the end-customer and user-centred approach in the same key position as it is in companies that are very mature in terms of customer-orientation.
Continuous development and improvement
Service development in regulated environments is usually a long-term and iterative process – rather than a one-off process – that continues throughout the service lifecycle. This kind of continuous long-term work also offers medical device designers the opportunity to improve the design and develop formative and summative evaluation methods, when more information is constantly gathered about the users and their needs. In addition to iterative design, medical devices require post-marketing surveillance, such as the continuous collection and analysis of user feedback, which brings even more input to user-oriented development.
At Solita, we have developed a model called RegProof®, in which the requirements for a medical device are integrated into agile and iterative development practices. At the same time, RegProof® aims to streamline the compilation of medical device documentation and break down the development of the service so that the service is close to being ready for release as often as possible. From a designer’s point of view, it is an interesting challenge to think how to optimally integrate the various formative evaluation service design methods and extensive summative usability testing into a faster release cycle. The journey to the release of a medical device is also a learning experience of transformation and changing operating methods for the designer.
The design of medical devices is challenged by the fact that even if their design and development methods could be optimised with the help of RegProof® or a similar agile development model, and even if the product development team together managed to implement a service that passes formative and summative evaluation, all significant changes to the medical device must be circulated through the notified body that oversees industry regulation before the service can be released. This takes its own, undetermined amount of time. Practical challenges related to notified bodies and supervising medical devices are currently being resolved at the level of the EU Commission, because the supervision infrastructure and its capacity have not yet had time to develop to meet the needs resulting from stricter regulation. This delays the entry of many medical devices onto the market and may force many manufacturers to narrow down their product portfolios. It is likely that the devices will be granted transition periods that are several years longer, though in some respects conditional. (More information about the transition periods and the challenges of MDR supervision, see e.g. Fimea’s website.)
Sustainable design and effectiveness
As I already mentioned, for many people the design of medical devices seems to be a meaningful design challenge in with a good purpose. That purpose is especially related to social sustainability, which is described, for example, in Goal 3 “Good health and well-being” of the UN Sustainable Development Goals.
Among other things, medical devices can help with communicable disease epidemics, as indicated by the COVID symptoms assessment in the Omaolo.fi service, or they can be used to promote mental health work, provide proactive healthcare or support lifestyle changes related to addictions. In addition, medical devices can also be used to improve the accessibility of services and regional equality or to reduce carbon dioxide emissions e.g. by enabling remote medical appointments or remotely controlled surgery.
By measuring the (social sustainability of) design, the impact of the design can be made visible. For example, the COVID symptoms assessment in Omaolo.fi and the Therapy Navigator service developed in the Terapiat etulinjaan project, have received Vaikuttavuusseura association’s impact award in recent years. Of these, Solita has been involved in the development of Omaolo and in investigatingwhether an assessment like the COVID19 symptom assessment could help anticipate the burden on the healthcare system during a pandemic.
Risk and quality requirements imposed on a medical device intent to ensure that the manufactured device will withstand time and predictable misuse, and that is patient safe and secure. Therefore, designing a medical device teaches the designer a lot about risk management, consideration of patient safety, quality and systematicity. The challenges with supervision that I already mentioned also teach us how to plan a service so that it stands the test of time as well as possible, without a need for unnecessary changes, but also so that it can be improved with the smallest possible increments that do not exceed the notified body’s significant change notification threshold.
Holisticityand working in a multi-skilled team
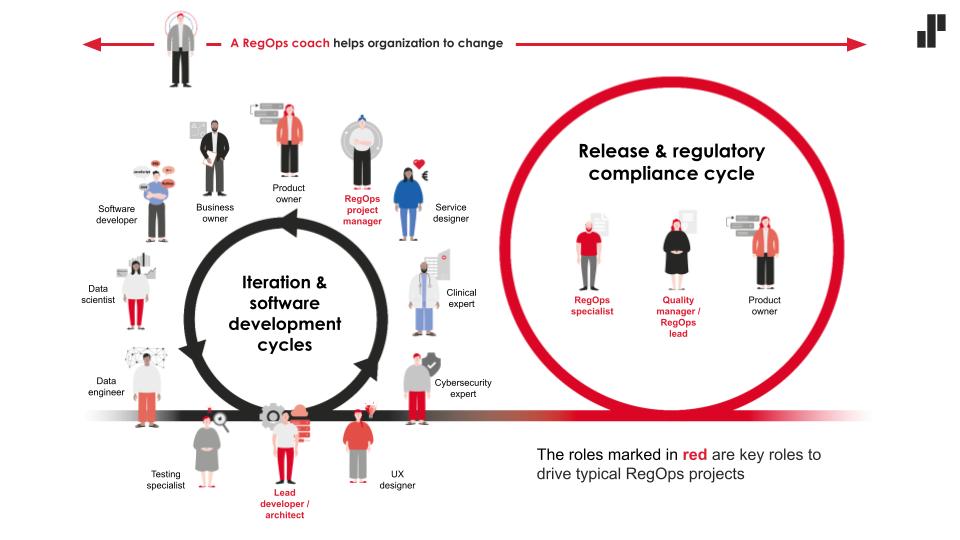
One of the designer’s important skills is to create convergence, i.e. integration and mutual understanding between people representing different backgrounds and skill palettes. The development of services in regulated environments requires in-depth multidisciplinary expertise, which teaches cooperation between designers, front- and back-end developers, data experts, clinicians, content producers, testers, and authors of user manuals, as well as with quality experts, risk experts and data protection experts.
If you are interested in being involved in planning better future health and well-being services, take a look at our vacant positions.